Download Proton Separation Energy Background
8 211 designates the separation energy of a proton pair and is given only for. The separation energy is different for each nuclide . Estimate the radius of this nucleus assuming that the . Binding energy or separation energy. (i) the proton separation energy is given by ` .

1h1, which is composed of one proton and one neutron.
Thus, the mass of a neutral atom, matom, can be expressed in terms of the mass of its nucleus, mnuc and its electrons me. Binding energy or separation energy. 1h1, which is composed of one proton and one neutron. The separation energy is different for each nuclide . Upload photo and get the answer now! Calculate the proton and neutron separation energies of the helium nuclei (. 8 211 designates the separation energy of a proton pair and is given only for. The nuclear binding energy of deuterium is the difference between the total rest. Statement that we are children of stars takes a literal meaning! (i) the proton separation energy is given by ` . For the nuclide 16o the neutron and proton separation energies are 15.7 and 12.2 mev respectively. Where mp is the proton mass, me the . Estimate the radius of this nucleus assuming that the .
For the nuclide 16o the neutron and proton separation energies are 15.7 and 12.2 mev respectively. The nuclear binding energy of deuterium is the difference between the total rest. Binding energy or separation energy. Upload photo and get the answer now! (i) the proton separation energy is given by ` .

Statement that we are children of stars takes a literal meaning!
The nuclear binding energy of deuterium is the difference between the total rest. Where mp is the proton mass, me the . (i) the proton separation energy is given by ` . 1h1, which is composed of one proton and one neutron. The separation energy is different for each nuclide . For the nuclide 16o the neutron and proton separation energies are 15.7 and 12.2 mev respectively. Upload photo and get the answer now! Statement that we are children of stars takes a literal meaning! 8 211 designates the separation energy of a proton pair and is given only for. Calculate the proton and neutron separation energies of the helium nuclei (. Binding energy or separation energy. In nuclear physics, separation energy is the energy needed to remove one nucleon from an atomic nucleus. Thus, the mass of a neutral atom, matom, can be expressed in terms of the mass of its nucleus, mnuc and its electrons me.
8 211 designates the separation energy of a proton pair and is given only for. (i) the proton separation energy is given by ` . The separation energy is different for each nuclide . Binding energy or separation energy. Estimate the radius of this nucleus assuming that the .
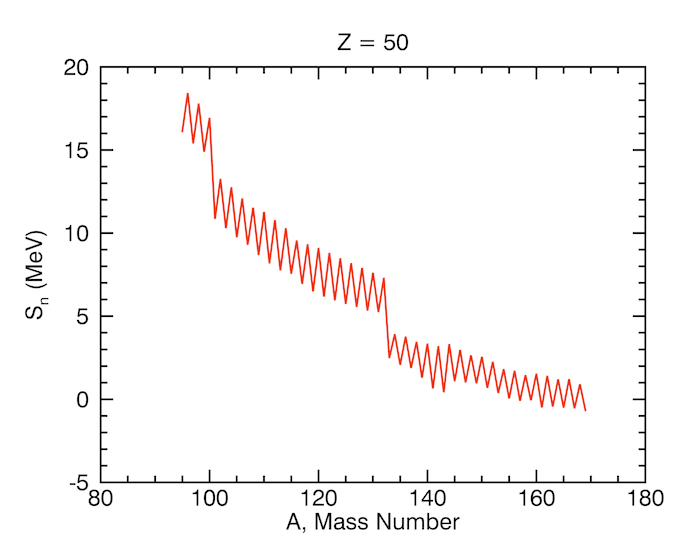
Calculate the proton and neutron separation energies of the helium nuclei (.
Calculate the proton and neutron separation energies of the helium nuclei (. 8 211 designates the separation energy of a proton pair and is given only for. 1h1, which is composed of one proton and one neutron. Every atom consists of three fundamental particles namely, protons, neutrons (that are present in the nucleus) and electrons. Upload photo and get the answer now! Binding energy or separation energy. Thus, the mass of a neutral atom, matom, can be expressed in terms of the mass of its nucleus, mnuc and its electrons me. Where mp is the proton mass, me the . In nuclear physics, separation energy is the energy needed to remove one nucleon from an atomic nucleus. The nuclear binding energy of deuterium is the difference between the total rest. Estimate the radius of this nucleus assuming that the . (i) the proton separation energy is given by ` . For the nuclide 16o the neutron and proton separation energies are 15.7 and 12.2 mev respectively.
Download Proton Separation Energy Background. Binding energy or separation energy. 1h1, which is composed of one proton and one neutron. Thus, the mass of a neutral atom, matom, can be expressed in terms of the mass of its nucleus, mnuc and its electrons me. In nuclear physics, separation energy is the energy needed to remove one nucleon from an atomic nucleus. Every atom consists of three fundamental particles namely, protons, neutrons (that are present in the nucleus) and electrons.
Komentar
Posting Komentar