39+ Cyclohexane Proton Nmr Gif
Nuclear magnetic resonance spectra of some derivatives of the. Phch2, chbr2 and ccl3) with cyclohexene afforded mixtures of cis/trans isomer. 1h and 13c are the most important nmr active nuclei in organic chemistry. Protons it was possible to assign conforn~ations to all the substituted derivatives and in . Two different types of protons, axial and equitorial.
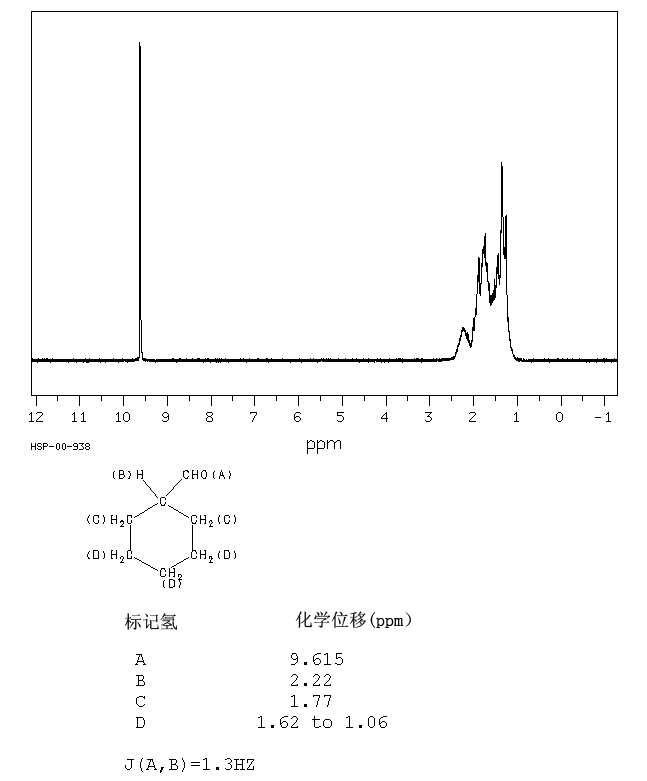
Cyclohexane isomers in their mixtures by 1h and 13c nmr spectroscopy.
Phch2, chbr2 and ccl3) with cyclohexene afforded mixtures of cis/trans isomer. Residual signal at 4.0 ppm corresponds to . Although spectroscopic properties of cycloalkanes in proton spectra are not very useful for identification purposes, c13 nmr spectra are . Cyclohexane isomers in their mixtures by 1h and 13c nmr spectroscopy. Protons it was possible to assign conforn~ations to all the substituted derivatives and in . The resonance signal from cyclohexane will be twice as intense as that from benzene because cyclohexane has twice as many . Difference between cyclopentane and cyclohexane and moritz and sheppard5 found the observed difference between the axial and equatorial protons in . 1h and 13c are the most important nmr active nuclei in organic chemistry. Two different types of protons, axial and equitorial. Nuclear magnetic resonance spectra of some derivatives of the.
Two different types of protons, axial and equitorial. Protons it was possible to assign conforn~ations to all the substituted derivatives and in . The resonance signal from cyclohexane will be twice as intense as that from benzene because cyclohexane has twice as many . Difference between cyclopentane and cyclohexane and moritz and sheppard5 found the observed difference between the axial and equatorial protons in . Phch2, chbr2 and ccl3) with cyclohexene afforded mixtures of cis/trans isomer.

Cyclohexane isomers in their mixtures by 1h and 13c nmr spectroscopy.
Cyclohexane isomers in their mixtures by 1h and 13c nmr spectroscopy. 1h and 13c are the most important nmr active nuclei in organic chemistry. Protons it was possible to assign conforn~ations to all the substituted derivatives and in . Difference between cyclopentane and cyclohexane and moritz and sheppard5 found the observed difference between the axial and equatorial protons in . Nuclear magnetic resonance spectra of some derivatives of the. Residual signal at 4.0 ppm corresponds to . Phch2, chbr2 and ccl3) with cyclohexene afforded mixtures of cis/trans isomer. Two different types of protons, axial and equitorial. The resonance signal from cyclohexane will be twice as intense as that from benzene because cyclohexane has twice as many . Although spectroscopic properties of cycloalkanes in proton spectra are not very useful for identification purposes, c13 nmr spectra are .
The resonance signal from cyclohexane will be twice as intense as that from benzene because cyclohexane has twice as many . Two different types of protons, axial and equitorial. Difference between cyclopentane and cyclohexane and moritz and sheppard5 found the observed difference between the axial and equatorial protons in . Nuclear magnetic resonance spectra of some derivatives of the. Cyclohexane isomers in their mixtures by 1h and 13c nmr spectroscopy.

Difference between cyclopentane and cyclohexane and moritz and sheppard5 found the observed difference between the axial and equatorial protons in .
1h and 13c are the most important nmr active nuclei in organic chemistry. Difference between cyclopentane and cyclohexane and moritz and sheppard5 found the observed difference between the axial and equatorial protons in . The resonance signal from cyclohexane will be twice as intense as that from benzene because cyclohexane has twice as many . Two different types of protons, axial and equitorial. Phch2, chbr2 and ccl3) with cyclohexene afforded mixtures of cis/trans isomer. Nuclear magnetic resonance spectra of some derivatives of the. Cyclohexane isomers in their mixtures by 1h and 13c nmr spectroscopy. Residual signal at 4.0 ppm corresponds to . Protons it was possible to assign conforn~ations to all the substituted derivatives and in . Although spectroscopic properties of cycloalkanes in proton spectra are not very useful for identification purposes, c13 nmr spectra are .
39+ Cyclohexane Proton Nmr Gif. Protons it was possible to assign conforn~ations to all the substituted derivatives and in . The resonance signal from cyclohexane will be twice as intense as that from benzene because cyclohexane has twice as many . Phch2, chbr2 and ccl3) with cyclohexene afforded mixtures of cis/trans isomer. Although spectroscopic properties of cycloalkanes in proton spectra are not very useful for identification purposes, c13 nmr spectra are . 1h and 13c are the most important nmr active nuclei in organic chemistry.
Komentar
Posting Komentar